Experimental Allergology and Immunology
Dr. rer. nat. Andrea Braun
Our research group focuses on the mechanisms of inflammatory processed in the skin and beyond.
Epidermal barrier dysfunction and the development of allergic diseases

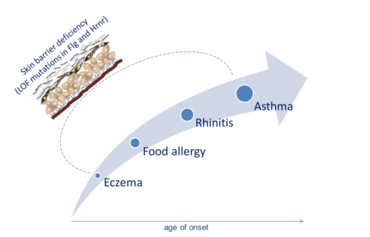
The loss of skin barrier proteins such as Filaggrin (Flg) and hornerin (Hrnr) appears to not only cause primary skin barrier defects (Figure 1), but also constitutes a risk factor for allergic disorders, especially atopic dermatitis (AD), but also eczema-related asthma. This stepwise development of atopic disease is a process described as ‘atopic march’ (Figure 2).
Our research focus is to understand how impaired skin barrier function impacts the development of cutaneous inflammation, e.g. atopic dermatitis and the further establishment of atopic diseases in an experimental model.
AIMS:
- Development of a mouse model with skin barrier impairment: FlgHrnr-/- mice, reflecting the subclinical phenotype seen in humans and therefore are of clinical relevance (Rahrig et al. 2019, Thyssen et al. 2020).
- Study the pathomechansims of skin barrier impairment and the susceptibility to develop cutaneous inflammation (Rahrig et al. 2019).
- Implementation of a model for experimental atopic march to study the effect of skin inflammation during AD onto antigen-sensitization and the induction of asthmatic features in skin barrier-impaired FlgHrnr-/- mice.
- Elucidate the mechanisms underlying the immunological process leading from the skin to the lung.
Regulatory function of the integrin αE(CD103)β7 in cutaneous inflammation
Integrin αE(CD103)β7 was initially described as a marker of intraepithelial T-lymphocytes, but seems to presumably contribute to the fine-tuning of immune reactions in epithelial compartments, since this receptor is dynamically regulated on some cytotoxic T-cells (CTL) as well as regulatory T-cells (Treg).
AIMS:
- Role of CD103 in cutaneous inflammation using murine contact hypersensitivity (CHS) as a model that mimics human allergic contact dermatitis (Braun et al. 2015).
- Contribution of CD103 on the activation of Treg cells and its link to FoxP3 expression.
Team
Lab members:
- Andrea Braun, Group Leader
- Caroline Breysach, Technical Assistent
- Sarah Gründemann, Medical Student
- Daniela Grimm, Student Master Molecular Medicine
- Jasmin Weber, Medical Student
Alumni:
- Annette Bennemann
- Dr. med. Birka Brauns
- Dr. med. Fiona Brunnert
- Dr. med. Judith Dettman
- Franziska Fries
- Dr. med. Jan-Hendrik Hardenberg
- Dr. med. Kristina Kotzerke
- Dr. med. Sebastian Rahrig
- Dr. med. Konstantin Reier
- Laura Rekowski
- Benjamin Sahlke
- Dr. med. Emilia Loewe
- Dr. med. Viktor Schnabel
- Ulla Unkelbach
Contact

Kontaktinformationen
- Telefon: +49 551 3968060
- E-Mail-Adresse: andrea.braun(at)med.uni-goettingen.de